MDIDS (Medical Device Interface Data Sheets)
Applying Medical Device Informatics to Enable Safe and Secure Interoperable Systems: Medical Device Interface Data Sheets
Goldman, Julian M. MD, Weininger, Sandy PhD; Jaffe, Michael B. PhD
doi: 10.1213/ANE.0000000000004251
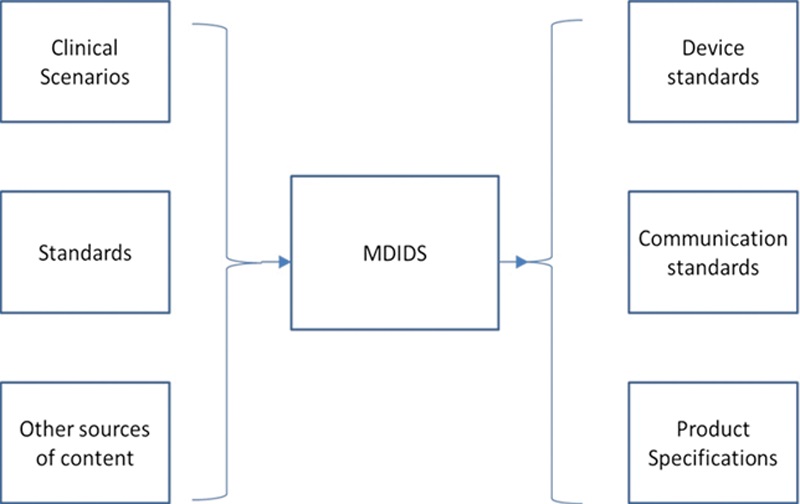
We have investigated multiple clinical scenarios in which connected medical devices are used as data sources, sensors, or actuators in distributed medical systems. This research has informed an approach to analyzing and reporting medical device interface capabilities and data elements which we capture and organize in a framework called “Medical Device Interface Data Sheets”. MDIDS can enable more complete, effective, and safe device integration for data access, device management, and device control.
These Medical Device Interface Data Sheets (MDIDS) have served as a reference for consensus medical device standards, manufacturers, researchers, and clinical organizations to facilitate interoperability and the safety of distributed systems.
We have develop device- and system-specific MDIDS
IV Pump MDIDS Poster from the Society for Technology in Anesthesia 2023 annual meeting:
“A Medical Device Information Data Sheet (MDIDS) to Support the Interoperability of Externally
Controllable Infusion Pumps for Tele-Critical Care“
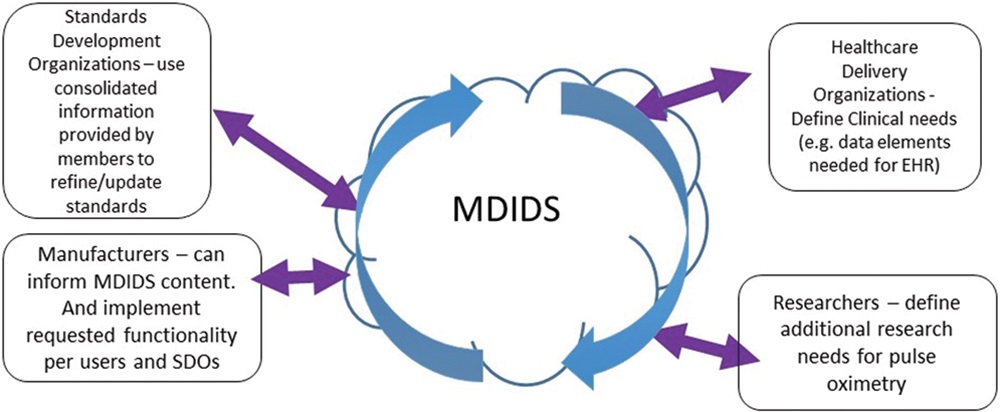
Clinical organizations are contributing interface capabilities that will support more effective device-to-EMR integration, innovative clinical care, and more effective clinical management.
Medical device manufacturers are contributing expertise to ensure completeness of the MDIDS.
Standards development experts are matching MDIDS data elements to existing standards and helping to identify standards gaps that must be addressed.
MDIDS include both “generic” and device-specific sheets.
Generic: The generic sheet includes device identification data such as Serial Number, FDA-defined Universal Device ID (UDI), software version numbers, and a description of the data encoding used for device-specific data elements. Other categories of data found in generic MDIDS are Patient Identification and Location information, as well as data about the Operating Conditions of the device and its Configuration.
We have developed example MDIDS for the Pulse Oximeter, Ventilator, Anesthesia Workstation, Defibrillator, and Dialysis Machine and. for closed-loop control applications.
Contact our team for information on how to develop MDIDS for your device or application.