CSR (Clinical Scenario Repository)

Capturing Essential Information to Achieve Safe Interoperability
Sandy Weininger, PhD, Michael B. Jaffe, PhD, Tracy Rausch, CCE, and Julian M. Goldman, MD
doi: 10.1213/ANE.0000000000001351
The CSR is a tool to elicit and document clinical situations in which improved technologies – especially those enabled through Integrated Clinical Environments – could improve patient safety and clinical care.
The CSR repository documents situations with potential or actual patient harm as well as hypothetical situations where a clinician or layperson sees room for improvement. No protected or confidential data is collected as part of the CSR. Solutions for these scenarios can be generalizable to create building blocks for interoperability to address many other healthcare needs.
The purpose of a clinical scenario is to provide a background and illustrate the need for the development of technical solutions, which can eventually be used by researchers, standards developers, regulators, and manufacturers to create innovative solutions for many intractable clinical problems and healthcare needs.
Based on a pilot deployment with physicians from the American Society of Anesthesiologists Committee on Patient Safety, the the CSR has been used to elicit “Good Ideas for Patient Safety” (GIPS).
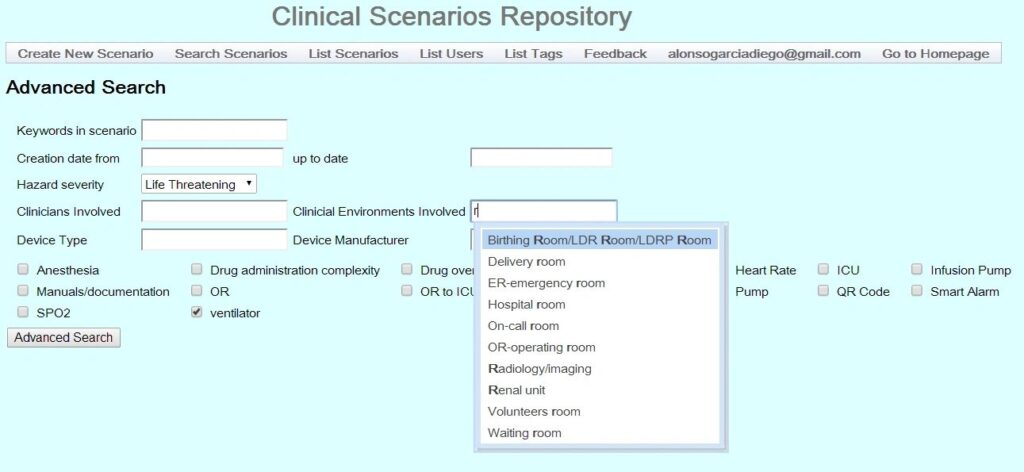
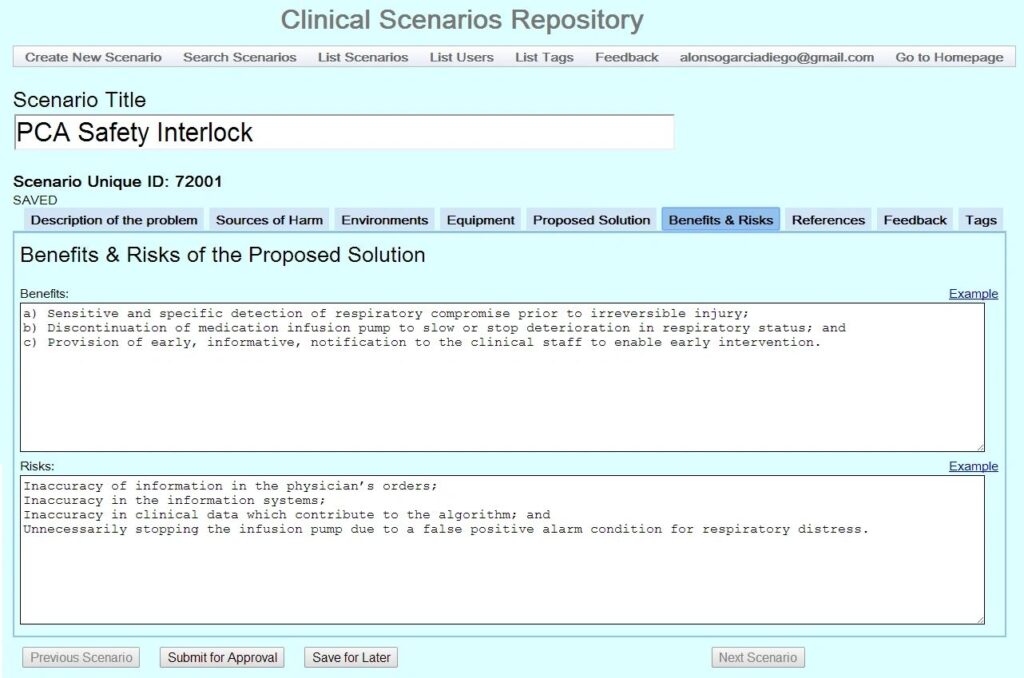
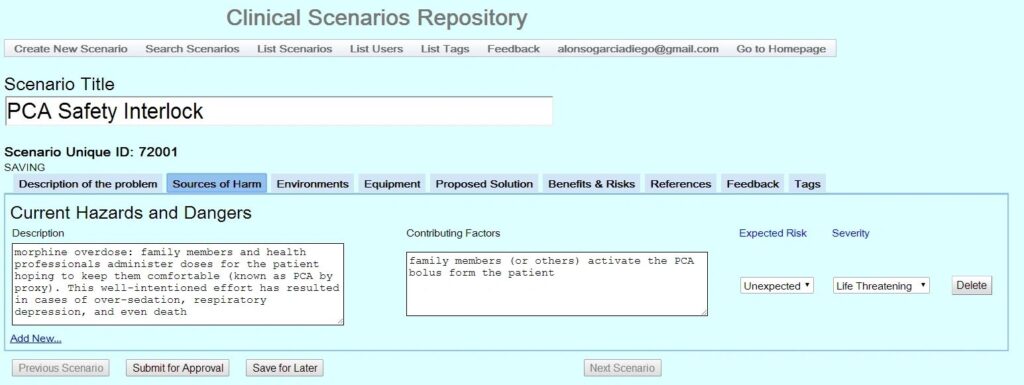
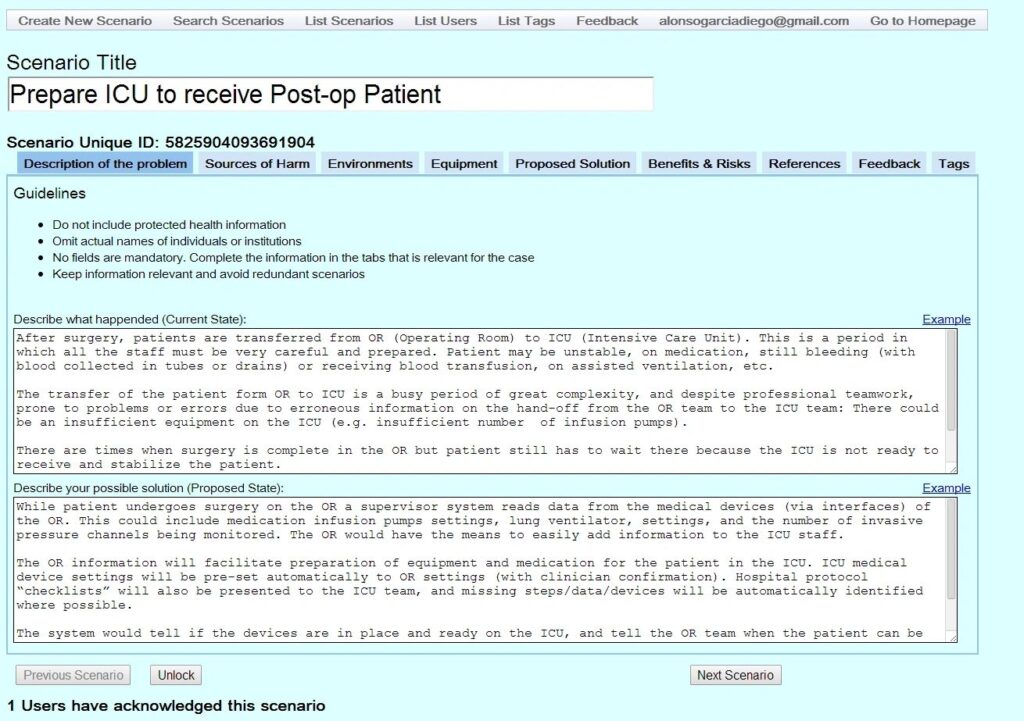
Click on the four thumbnails below to see examples of a prior version of the CSR. Stay tuned for updated screen shots.